How EMS-Driven Precision PCBA Ensures Reliability in Medical Devices

Reliability in medical electronics is not probabilistic but is engineered to operate within defined limits over time, load, and environment, established through controlled tolerances, component derating, and robust system architectures.
Devices used in monitoring, diagnostics, and patient care must deliver consistent, repeatable performance across extended duty cycles without failure, enforced through rigorous validation and disciplined process control across the product lifecycle.
In practice, this translates into failure rates approaching parts per million (PPM) levels, with high-reliability assemblies often targeting <50 PPM defect rates in production environments. For critical subsystems, even lower thresholds are enforced through process control and validation.
Achieving this level of reliability is not a function of design alone. It is a function of how consistently that design is manufactured, specifically at the level of PCB assembly (PCBA).
Reliability Targets in Medical Electronics
Medical devices operate under conditions where both functional accuracy and temporal stability are required.
Typical expectations include:
- 24/7 operational stability for monitoring systems
- Signal accuracy within tight tolerances (often <1–2% deviation depending on application)
- Multi-year lifecycle reliability under repeated thermal and electrical stress
- Zero tolerance for intermittent faults in critical systems
Unlike consumer electronics, where failures are often binary, medical device failures are frequently latent or intermittent, making them harder to detect and more critical to prevent.

Where PCBA Impacts Reliability Quantitatively
1. Solder Joint Integrity and Field Failure Rates
Industry data indicates that over 60% of electronic failures in high-density assemblies can be traced to solder joint defects.
Failure modes include:
- Micro-cracks due to thermal cycling
- Voiding in BGA packages affecting heat dissipation
- Cold joints leading to intermittent connectivity
For medical-grade assemblies, voiding thresholds are typically controlled below ~20–25% in critical pads, with tighter internal targets depending on application.
2. Placement Accuracy and Signal Integrity
Modern medical devices, especially wearables and compact diagnostic systems, rely on high-density PCB layouts.
Typical SMT placement tolerances:
- ±25–50 microns for fine-pitch components
Deviation beyond this can lead to:
- Impedance mismatch in high-speed signals
- Increased electromagnetic interference (EMI)
- Unreliable contact points in miniature packages
In wearable medical devices such as smart monitoring watches or flexible dental electronics, these tolerances directly affect sensor accuracy and data consistency.
3. Thermal Profile Control and Component Stress
Reflow soldering introduces thermal exposure that can affect both components and interconnect reliability.
Improper profiling can result in:
- ΔT (temperature variation) across board >10–15°C, leading to uneven soldering
- Intermetallic compound (IMC) instability
- Long-term fatigue failure under repeated heating cycles
Medical-grade PCBA typically enforces tightly controlled thermal gradients to ensure uniform joint formation and minimize stress.
4. Inspection Depth vs Defect Escape Rate
Inspection systems directly influence defect escape rates, which are critical in medical applications.
Typical detection layers include:
- AOI (surface-level defects)
- X-ray inspection (hidden joints such as BGAs, QFNs)
- Functional validation
Without X-ray validation, hidden solder defects can account for up to 20–30% of undetected failures in complex assemblies.
5. Functional Testing and Reliability Validation
Beyond assembly, boards must be validated under electrical and operational stress.
Medical-grade validation often includes:
- In-Circuit Testing (ICT) for component-level verification
- Functional Testing (FCT) for system behavior
- Burn-in cycles (24–72 hours or more) to expose early-life failures
This reduces the probability of infant mortality failures, which are statistically more likely in the early lifecycle phase.
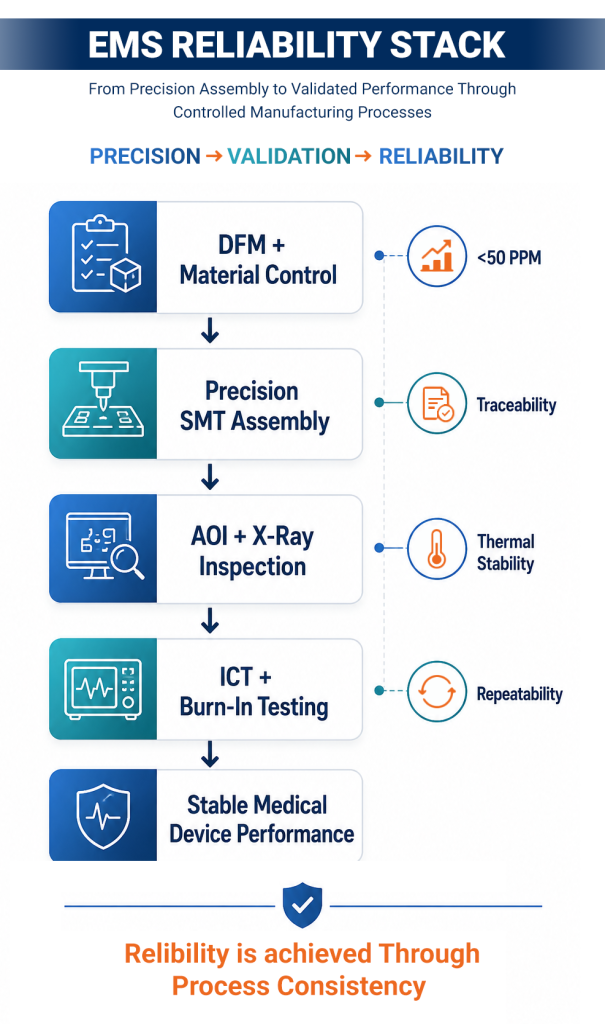
How EMS Ensures the Reliability
Achieving consistent reliability at a scale requires more than capability. It requires disciplined execution across manufacturing systems, where EMS providers play a critical role.
Process Control
Controlled SMT processes, thermal profiling, and standardized assembly conditions ensure repeatability across production batches.
Traceability
End-to-end tracking of components, materials, and process parameters enables compliance, accountability, and efficient root-cause analysis.
Inspection Systems
Multi-layer inspection, including AOI and X-ray, minimizes defect escape rates and ensures structural integrity in complex assemblies.
Validation Frameworks
Structured validation through ICT, FCT, and burn-in testing ensures functional reliability and early detection of potential failures before deployment.
Together, these systems translate design intent into consistent manufacturing outcomes across volume production
Performance in Real-World Conditions
The reliability established at the assembly stage is ultimately validated under real-world operating conditions.
Medical devices are rarely used in ideal environments, and assembly-level variations often become more pronounced when exposed to external stress over time.
Typical stress factors include:
- Thermal cycling (0°C to 50°C or higher depending on use case)
- Humidity exposure affecting insulation resistance
- Continuous operation leading to material fatigue
To mitigate this, precision PCBA integrates:
- Conformal coatings for protection
- Controlled assembly environments
- Traceability systems for component and process tracking
The effect of these controls becomes more apparent when observed across different medical device categories, where environmental exposure and operational demands vary significantly.
Applied Context: Real Device Categories
Precision PCBA requirements vary across device types, but the underlying reliability principles remain consistent as assembly precision determines how well a device withstands its operating environment over time.
Wearable Medical Devices (e.g., Smart Watches, Dental Wearables)
- Ultra-compact layouts with high component density
- Sensitivity to motion, temperature, and user environment
- Requirement for stable sensor output over time
Environmental & Air Quality Monitoring Devices
- Continuous sensing with calibration stability
- Exposure to particulate matter and environmental variation
- Need for long-term drift control
In air quality monitoring systems designed for continuous operation, assembly precision directly influences calibration retention, as prolonged environmental exposure can amplify minor variations into measurable sensor drift over time.
Critical Medical Equipment Subsystems
- Zero tolerance for intermittent faults
- High-reliability interconnects and power stability
- Strict validation and traceability requirements
In high-reliability assemblies used within critical equipment, where PCBAs function as core subsystems, consistency across batches becomes essential, with process control and validation frameworks ensuring stable performance under continuous operation.
In each case, assembly precision directly affects data accuracy, device stability, and lifecycle performance.
Precision as a Statistical Outcome, not a claim
Across device categories and operating environments, precision in PCBA is not defined by capability alone; it is defined by process consistency over volume.
What begins as assembly-level control ultimately manifests as measurable outcomes across production and field performance.
Key indicators include:
- Defect rates (PPM levels)
- Yield stability across batches
- Repeatability of electrical performance
- Long-term field reliability data
For medical electronics, success is measured not by whether a device works once, but by whether it continues to operate within defined parameters across its entire lifecycle.
Reliability, Defined at Assembly
In high-reliability medical systems, design defines intent—but assembly determines whether that intent holds under real-world conditions.
PCB assembly introduces the physical variables that govern stability, repeatability, and long-term performance.
Reliability, therefore, is not an inherent attribute of design but an outcome of disciplined manufacturing execution. Precision PCBA is a controlled engineering process that ensures medical devices consistently meet defined reliability thresholds, not just in isolated builds but across every unit at scale.
